The Government is launching a fresh drive to encourage people to accept a coronavirus vaccine amid continuing reluctance among some groups to have the jab.
Ministers are confident they will achieve their UK-wide target of getting an offer of a vaccine to those most at risk from the virus – including all over 70s – by Monday’s deadline.
Health Secretary Matt Hancock said he hoped a combination of vaccines and new treatments will mean Covid-19 could be a “treatable disease” by the end of the year.
Mr Hancock issued a direct appeal to anyone over 70 who has still not had the jab to contact the NHS over the weekend to book an appointment.
“I am determined that we protect as many of our country’s most vulnerable people from this awful disease as soon as possible,” he said. “Vaccines are the way out of this pandemic.”
Overall, uptake of the vaccine has been high, with the Department of Health and Social Care (DHSC) reporting a 93% take-up rate among the over 75s in England.
The DHSC is now seeking to work with community organisations and charities in England to address the concerns that are making some reluctant to get the jab, while seeking to dispel “myths” circulating on social media.
Around 30 ministers are taking part in visits and virtual meetings, including Home Secretary Priti Patel and Vaccines Minister Nadhim Zahawi.
“We recognise that some groups feel more hesitant about getting a jab, or have more barriers, both physical and mental, preventing them from accessing one when it’s offered,” Mr Zahawi said.
Mr Hancock, meanwhile, expressed the hope that coronavirus will become “another illness that we have to live with” like flu.
“I hope that Covid-19 will become a treatable disease by the end of the year,” Mr Hancock told The Daily Telegraph.
People who are aged 70 and over who have not yet had their #COVIDVaccine, and would like to, can contact the NHS to arrange their appointment.
For all the latest on booking a COVID-19 vaccine, visit https://t.co/usB1YdJfNn pic.twitter.com/lbDXyrFvsw
— NHS England and NHS Improvement (@NHSEngland) February 12, 2021
“If Covid-19 ends up like flu, so we live our normal lives and we mitigate through vaccines and treatments, then we can get on with everything again.”
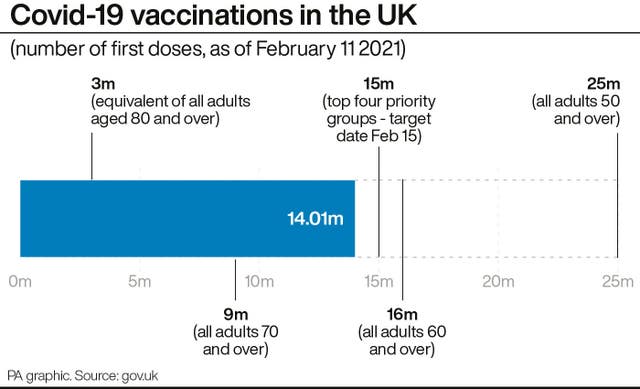
The move comes as it was announced on Friday that more than 14 million across the UK have now received their first dose of one of the approved vaccines.
NHS England said the top four priority groups in England – people aged 70 and over, care home residents and staff, health and care workers and clinically extremely vulnerable patients – “have now been offered the opportunity to be vaccinated”, while Wales said those groups had been reached.
NHS England said people aged 65 to 69 can now get a vaccine if GPs have supplies, while Welsh First Minister Mark Drakeford said they had already begun contacting some over 50s.
In Northern Ireland, the Department of Health is offering everyone over 65 a vaccine by the end of February as it works its way through priority groups four and five, although it is expected to help the UK meet its overall target.
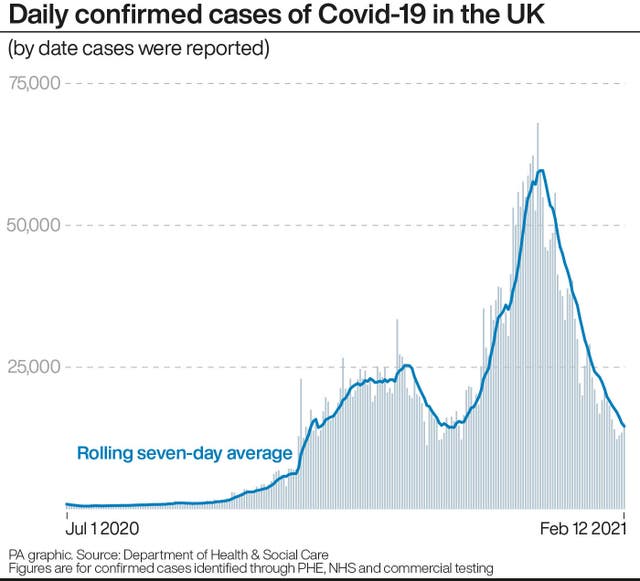
It comes as the reproduction number, or R value, for coronavirus is now estimated to be between 0.7 and 0.9 across the UK.
This is the first time since July that R has been this low, indicating that lockdown restrictions are having an impact and the epidemic is shrinking.
As of February 9, the latest date for which figures are available, there were 25,621 patients in hospital in the UK with Covid-19 – down from a peak of over 39,000 in mid January.
The figures are likely to intensify the pressure on Mr Johnson from some Tory lockdown-sceptics to to begin easing restrictions and re-opening the economy.

However, scientists advising the Government continue to urge caution, arguing that case numbers remain too high to allow any significant easing of the controls.
Meanwhile, the DHSC said treatments for Covid-19 will soon be fast-tracked through the UK’s clinical trial system, meaning they could be available on the NHS in months rather than years.
The Government, which currently funds phase 2 and 3 trials such as the Recovery trial, which brought dexamethasone and tocilizumab to the NHS, has awarded multimillion-pound funding to a phase 1 clinical trial platform.
Phase 1 trials, usually arranged by researchers, are the earliest stage of human trials that ensure treatments are safe and show a signal of benefit in treating a disease.
The funding has been awarded to expand the Agile clinical trial platform and will allow for the progress of cutting-edge treatments for Covid-19 through all three clinical trial phases in the UK – a streamlined process that is hoped to protect the supply chain.