The EU drugs regulator has authorised Pfizer’s coronavirus vaccine for use on children aged five to 11, clearing the way for jabs to be administered to millions of primary school pupils amid a new wave of infections sweeping across Europe.
It is the first time the European Medicines Agency has cleared a Covid-19 vaccine for use in young children.
The agency said it “recommended granting an extension of indication for the Covid-19 vaccine Comirnaty to include use in children aged five to 11″.
‼️ EMA recommends approval of BioNTech/Pfizer’s #COVID19vaccine, Comirnaty, for children aged 5 to 11.
In this population, the dose of #Comirnaty will be lower than that used in people aged 12 and above.
Read the full press release: https://t.co/uqqBAklVvl pic.twitter.com/NZQhli4SDl
— EU Medicines Agency (@EMA_News) November 25, 2021
After evaluating a study of the vaccine in more than 2,000 youngsters, the EMA estimated it was about 90% effective in preventing symptomatic Covid-19 in young children, and said the most common side effects were pain at the injection site, headaches, muscle pain and chills.
At least one country facing spiking infections did not wait for EMA approval. Authorities in Austrian capital Vienna have already started vaccinating the age group.
Europe is at the epicentre of the pandemic and the World Health Organisation has warned the continent could see deaths top two million by the spring unless urgent measures are taken.
The EMA approval for the vaccine developed by Pfizer and German company BioNTech has to be rubber-stamped by the European Commission before health authorities in member states can begin administering jabs.
Earlier this week, German health minister Jens Spahn said shipping of vaccines for younger children in the EU would begin on December 20.
The US signed off on Pfizer’s vaccine for children earlier this month, followed by other countries including Canada.
Pfizer tested a dose that is a third of the amount given to adults for primary school-age children. Even with the smaller jab, children who are five to 11 years old developed antibody levels as strong as teenagers and young adults getting the regular-strength vaccines, Pfizer said in September.
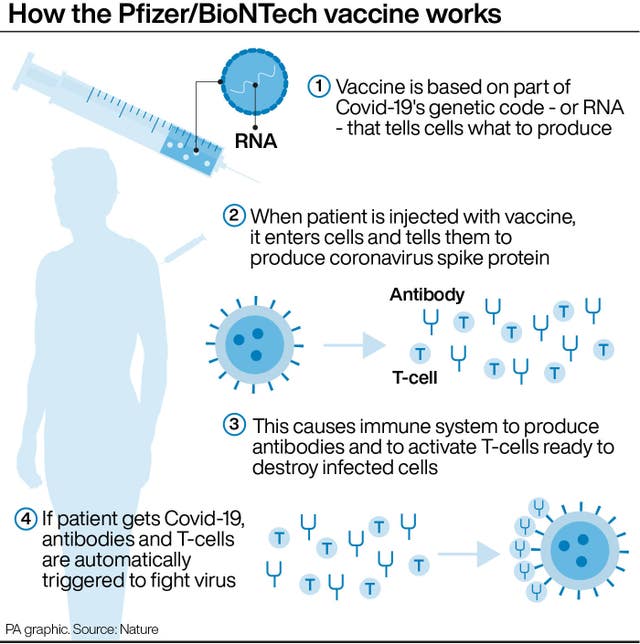
US officials noted that Covid-19 has caused more deaths in children in the five to 11 age group than some other diseases, such as chicken pox, did before children were routinely vaccinated.
Earlier this month, the EMA said it began evaluating the use of Moderna’s Covid-19 vaccine for children aged six to 11, and estimated a decision would be made within two months.
Although children mostly only get mild symptoms, some public health experts believe immunising them should be a priority to reduce the virus’s spread, which could theoretically lead to the emergence of a dangerous new variant.
Researchers disagree on how much children have influenced the course of the pandemic. Early research suggested they did not contribute much to viral spread, but some experts say children played a significant role this year spreading variants such as Alpha and Delta.
In a statement this week, the WHO said that because children and teenagers tend to have milder Covid-19 disease than adults, “it is less urgent to vaccinate them than older people, those with chronic health conditions and health workers”.
It has appealed to rich countries to stop immunising children and asked them to donate vaccines to poor countries who have yet to give a first dose to health workers and vulnerable populations.