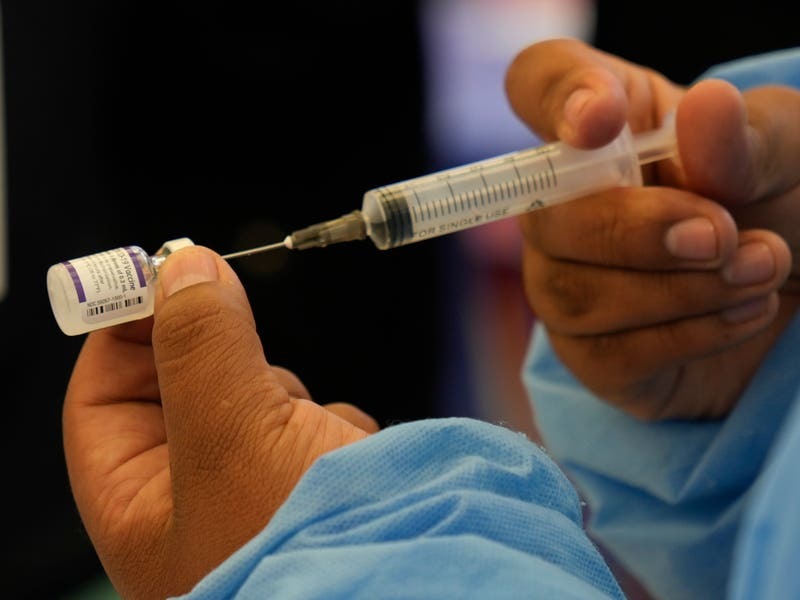
The US moved a step closer to expanding Covid-19 vaccinations for millions more children as government advisers endorsed the Pfizer vaccine for kids aged between five and 11.
A Food and Drug Administration (FDA) advisory panel voted unanimously, with one abstention, that the vaccine’s benefits in preventing Covid-19 in that age group outweigh any potential risks.
That includes questions about a heart-related side effect that has been very rare in teens and young adults despite their use of a much higher vaccine dose.
While children are far less likely than older people to get severe Covid-19, ultimately many panellists decided it is important to give parents the choice to protect their youngsters — especially those at high risk of illness or who live in places where other precautions, like masks in schools, are not being used.
The FDA is not bound by the panel’s recommendation and is expected to make its own decision within days.
If the FDA concurs, there is still another step: Next week, the CDC will have to decide whether to recommend the jabs and which youngsters should get them.
Full-strength vaccines made by Pfizer and its partner BioNTech already are recommended for everyone 12 and older but paediatricians and many parents are clamouring for protection for younger children.
The extra-contagious delta variant has caused an alarming rise in paediatric infections, and families are frustrated with school quarantines and having to say no to sleepovers and other rites of childhood to keep the virus at bay.
In the five-to-11 age group, there have been over 8,300 hospitalisations reported, about a third requiring intensive care, and nearly 100 deaths.
States are getting ready to roll out the jabs — just a third of the amount given to teens and adults — that will come in special orange-capped vials to avoid dosage mix-ups.