A small number of reports associated with blood clots have been notified to Irish regulators following vaccination with AstraZeneca.
They were not as serious as those described in Norway, which led to a temporary halt on the drug’s use in Ireland, and the number involved was extremely low, the country’s Health Products Regulatory Authority (HPRA) said.
The European Medicines Agency (EMA) has initiated an urgent review of all clotting events occurring with the vaccine to determine if there is a possible safety risk.
The National Immunisation Advisory Committee has recommended the temporary deferral of the administration of the COVID-19 Vaccine AstraZeneca® (14 March 2021)
Questions & Answers document now available from our website https://t.co/Y45xvKKG20 #COVIDVaccine #HoldFirm
— HSE National Immunisation Office (NIO) (@HSEImm) March 14, 2021
A statement from the HPRA said: “To date, the HPRA has received a small number of reports associated with blood clots following vaccination with the AstraZeneca vaccine.
“However, it has not received any reports of the nature of those described by the Norwegian Medicines Agency.
“We will continue to monitor national reports very closely and continue to encourage the reporting of any suspected side effect following vaccination with a Covid-19 vaccine.”
It said there was no indication that vaccination was the cause of these events and there may be alternative, unrelated explanations for their occurrence.
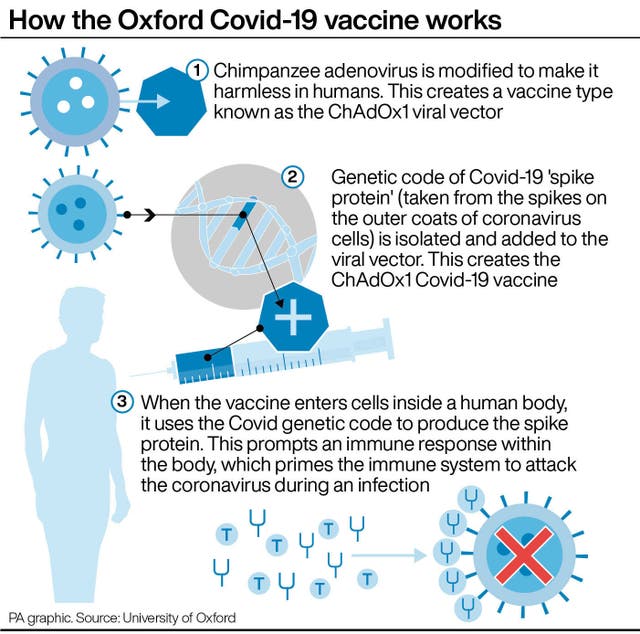
The preliminary assessment of similar events published by the EMA on March 11 found the number of clotting events in vaccinated people was no higher than the number seen in the general population.
As of March 10, 30 cases were reported in the EMA’s database of side effects following vaccination of close to five million people in the European Economic Area.
A thorough analysis of all relevant reports would be performed, including those newly notified from Norway, authorities said.
He told RTE’s This Week programme that there had been no reports of similar clotting events in Ireland akin to those seen in Norway.
“It’s very important to reassure people that there is no evidence of a cause and effect at this point,” he said.
“We have a safety signal and when we get those we have to act and proceed on the basis of a precautionary principle. So hopefully, as this week goes on, we’ll get more reassuring data from the EMA and we can recommence the programme.
“It may be nothing, we may be overreacting, and I sincerely hope that in a week’s time we are accused of being overcautious.”